iTind – natural and safe treatment for prostate enlargement
Benign prostatic hyperplasia (BPH) is a common condition that affects an increasing number of men as they age, causing difficulties with urination, frequent visits to the toilet and sleep disturbances, among other things. Although BPH is not cancer, it can significantly reduce quality of life. A modern and minimally invasive solution to this problem is the iTind procedure – a clinically proven method that safely and effectively relieves the symptoms of prostate enlargement without the need for daily medication or surgery.
BPH at age 60
BPH at age 85
Benign prostatic hyperplasia (BPH) – a complete guide
Benign prostatic hyperplasia (BPH) is a medical term describing prostate enlargement, a condition that can affect urination. It is a very common condition. Many men worry that BPH can turn into prostate cancer. This is not true. BPH is not cancer and cannot turn into cancer. However, prostate enlargement can interfere with sleep, social life and have a serious impact on quality of life.
How to recognise BPH? The most common symptoms
An enlarged prostate can put pressure on the urethra, the tube through which urine flows. This can affect urination and cause:
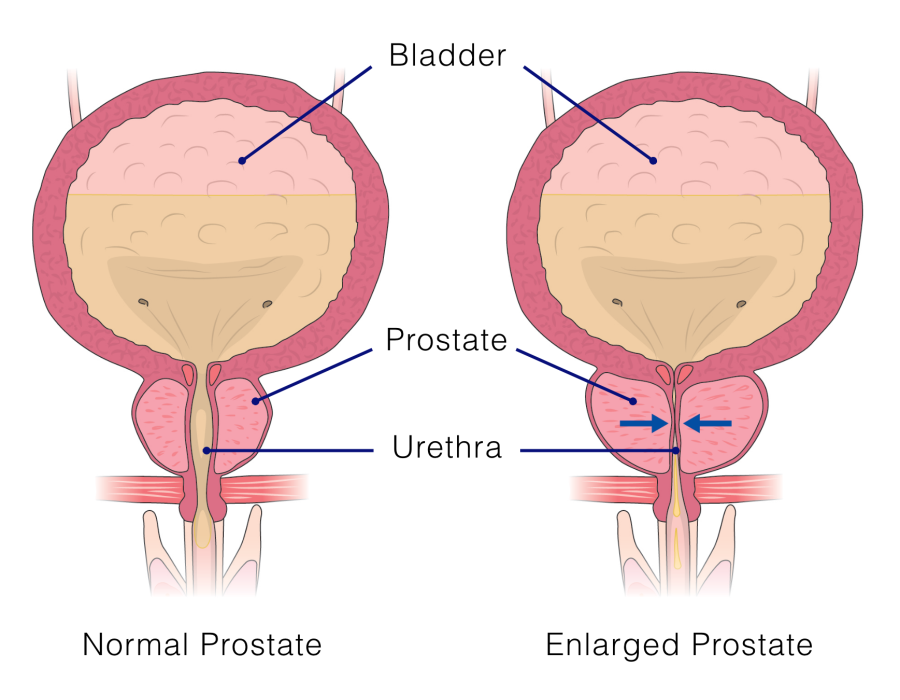
iTind – an innovative solution for men with prostate problems
iTind is a minimally invasive and clinically proven treatment that provides fast and effective relief from BPH symptoms.
The procedure involves implanting a small device into the prostate for 5–7 days, after which it is completely removed. Once implanted, iTind expands and exerts gentle pressure, remodelling the tissue and creating a wider channel through which urine can flow.
The iTind procedure is an ideal alternative to prescription drugs or invasive surgical procedures. You should consider iTind if you do not want to take tablets every day or if you have tried standard pharmacological treatments and are not satisfied with the level of symptom relief or side effects.

Effects of iTind – what can you expect?
The procedure can be performed under light sedation or local anaesthesia, usually takes less than 30 minutes, and does not require heating or removal of prostate tissue.
iTind is designed not only to effectively relieve symptoms, but also to provide peace of mind.
Benefits of using iTind:
What can I expect from the iTind procedure?
The iTind procedure is a minimally invasive procedure performed by a urologist that allows you to quickly return to your daily activities. The device remains in the prostate for several days, after which it is safely removed and the patient gradually experiences relief from the symptoms of prostate enlargement.

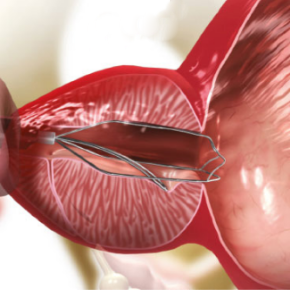
Introduction of iTind
The iTind procedure will be performed by a urologist in an operating theatre or outpatient clinic. The urologist may give the patient a mild sedative and local anaesthetic.
A thin tube with a miniature camera will be inserted into the urethra to determine the placement of the iTind device. The urologist will then place the iTind device in the prostate.
Once the device is in place, the patient should be able to urinate freely and will be discharged home. A catheter is not usually required.
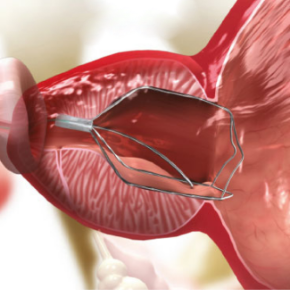
Implantation period
Over the next 5–7 days, you can return to most normal activities, depending on how you feel. You may experience pain in your lower abdomen and discomfort when sitting. You may feel a more frequent need to urinate and a greater sense of urgency. There may also be blood in your urine. These are normal reactions.
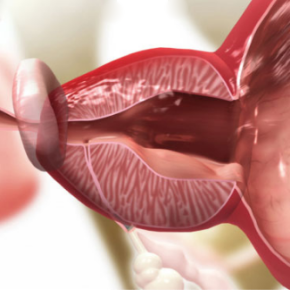
Removal of iTind
After 5–7 days, your urologist will completely remove the iTind device using a flexible silicone catheter. You can resume normal activities 1–2 days after removal.
A quick return to everyday life!